I was a chemical engineer on the one hand, and then I would be exposed to medicine. I would have two different disciplines. They were so different. You'd think ‘Well, you could combine them,’ and that would give me ideas that nobody at that time had — because nobody else had that kind of background.

Robert Samuel Langer, Jr. was born and raised in Albany, New York. His father was a businessman who ran a billiard parlor in nearby Troy, New York, and then a liquor store in Albany. As a youngster, Robert enjoyed magic tricks, and when he was given a Gilbert chemistry set at age 11, he delighted mixing the colors and observing the chemical reactions. Soon he was making rubber and synthesizing simple plastics. At the Milne School in Albany, he excelled in math and science, and he was encouraged by his family to study engineering in college. As a freshman at Cornell University, he most enjoyed his chemistry class, and decided to major in chemical engineering. He graduated from Cornell in 1970 and pursued graduate studies at Massachusetts Institute of Technology (MIT). As Langer was working towards a doctorate, the U.S. passed through a severe gasoline shortage. American industry was eagerly searching for ways to increase fuel efficiency, and chemical engineers were in high demand. Most of Langer’s classmates headed for careers in the energy industry. Langer himself received 20 offers from oil companies, including Shell and Chevron, and four from Exxon alone, but he couldn’t interest himself in fuel technology. While studying in Cambridge, he had developed a chemistry and math curriculum for inner city school children, and he was still looking for a career where he could help others more directly than he felt he could do in the oil industry.

Langer applied for research positions at 40 colleges and universities without hearing back from any of them. Nine research grant proposals he wrote were rejected. Then he wrote to hospitals and medical schools to see if they could use a chemical engineer, but he received no offers. Finally, a friend in Cambridge recommended he contact Dr. Judah Folkman, a Harvard professor who was also chief of surgery at Boston’s Children’s Hospital. At the time, very few chemical engineers were working in surgical labs, but Dr. Folkman was pursuing an unorthodox approach to cancer research and was willing to employ postdoctoral fellows without conventional medical or biological training. Folkman believed that the spread of cancer and the growth of tumors could be controlled if angiogenesis, the process by which new blood vessels are created, was arrested. He set Langer the task of finding substances to inhibit the creation of new blood vessels. While in Folkman’s lab, Langer pursued a second avenue of research. He was searching for polymers that would permit the gradual timed release of medication within the body.

Other scientists had considered this idea and abandoned it as impossible, but Langer believed it could be done and undertook the painstaking process of eliminating possible formulae. In the first two years, he tried more than 200 variations, before finding a technique for modifying polymers that would slowly release the molecules for drug delivery. The same technique facilitated the testing of substances for inhibiting angiogenesis.

Two years into this work, Langer was called on to give a talk about his research for a conference of polymer chemists and engineers, but he was met with incredulity. He fared no better when he submitted research proposals to the National Institutes of Health (NIH). His first nine research grant applications were turned down. From his first publication on angiogenesis, nearly 28 years would elapse before the Food and Drug Administration approved the first anti-cancer drug based on this work.
|
|
After completing his postdoctoral work, Langer applied for positions in chemical engineering departments, but met a cool response. While the biologists at NIH had felt that, as a chemical engineer, Langer couldn’t know anything about biology, much less cancer, the chemical engineering faculty at most of the universities where he applied believed his research belonged in the biology department or the medical school. Finally he was offered a post in the Nutrition and Food Science program at MIT, but in his first years at MIT, even this position was precarious.
Since there was little interest in his work in the academic and institutional worlds, he set out to interest the private sector. He began writing patents for his discoveries, in the hope that he could license them to pharmaceutical companies that would finance their further development and bring them to market, where they could benefit patients directly.

His first patents concerned polymer systems for controlled release of macromolecules. Again, he had difficulty persuading others of the value of his ideas. The reviewers at the patent office didn’t believe controlled release would work. His application was turned down five times between 1976 and 1981, and the attorney handling his application advised him to give up. When Langer demonstrated the effectiveness of his work, the patent office replied that his work was nothing new. In the end, to prove his work was original, he procured affidavits from the authors of a 1979 paper who had flagged his approach as being far out of the mainstream.
With patents in hand, Langer applied to NIH for a grant to support developing biodegradable polymers for the delivery of brain cancer drugs, but the reviewers did not believe such polymers could be synthesized. Langer’s grant applications were rejected many times in the years to come.

Once he had secured his patents, Langer still had difficulty finding interested companies to pursue his ideas. In 1983, International Minerals & Chemicals licensed one of his patents, and hired him as a consultant for a project using polymers to deliver animal hormones. The following year, Eli Lilly & Co. signed him for a similar project with human hormones, but when the first trials were unsuccessful, these large businesses quickly lost interest. In 1985, Langer made a deal with a much smaller company, Nova Pharmaceuticals of Baltimore, to license one of Langer’s patents for the delivery of brain cancer treatment. Unlike the larger companies, Nova was fully committed to innovation. Langer realized that working with small, narrowly focused enterprises was the best way to advance his ideas in the marketplace.

Even before his success with Nova, Langer had been approached by venture capitalists about starting a company of his own, and in 1987, he and a colleague, Alexander Klibanov, founded Enzytech. The company produced and brought to market a microsphere drug delivery system. Products based on the principles Langer developed have since been used to treat alcoholism, narcotic addiction, diabetes and other diseases. Enzytech was later merged with another company to form Alkermes.
Jay Vacanti, a surgeon at Massachusetts General Hospital, interested Langer in combining three-dimensional synthetic polymer scaffolds with living cells to create new tissues and organs in the laboratory. At the time, Langer was unable to get government grants for this research, so Langer began to look for commercial funding. In 1988, he started a second company, Neomorphics, to produce biocompatible materials for tissue growth. Like Enzytech, Neomorphics was later acquired by larger concerns, Advanced Tissue Sciences and Smith & Nephew. As his ideas achieved success in the marketplace and acceptance in academia, Langer’s position at MIT became more secure and he was given a full professorship.

Whereas other researchers tried to find medical applications for existing plastics, Langer identified the qualities he was looking for and synthesized new materials to meet the need, creating a new family of biodegradable polymers, the polyanhydrides. In 1992, Langer founded Focal to produce biodegradable materials for sealants and for the prevention of surgical adhesions. The following year saw the creation of two new enterprises: Enzymed, which produced combinatory pharmaceuticals; and Acusphere, which developed imaging agents with porous microsphere technology. The first two of these concerns were also merged with larger companies, and the third became a publicly traded company, continuing Langer’s winning streak in the business world.

In the 1990s, Robert Langer established a new model of research and development. If a discovery in his laboratory is a genuine breakthrough that offers the possibility of multiple applications, and an exclusive patent can be obtained, and if students or postdoctoral fellows of Langer’s have worked with the technology for five years or more, Langer encourages them to start a new company to develop the idea if they are interested. By collaborating with his students in the founding of these businesses, Langer mentors them as both scientists and entrepreneurs. At first, this practice was controversial, but these startups have created new treatments for cancer, heart disease and other deadly ailments, while providing employment to thousands.
After the initial startup period, Langer often withdraws from the enterprise, although sometimes he remains on board as a paid advisor or board member. When a discovery is licensed from MIT, the profits are split between the department, the university and the inventors. MIT scientists are permitted to take equity stakes in the companies that employ their discoveries, but they cannot own shares in businesses that provide research grants to their laboratories, and they cannot serve as executives of outside firms, although they may serve as paid advisors.

In 1996, the FDA approved the use of Langer’s controlled-release drug delivery system for the treatment of brain cancer. In the next few years, Langer and his students were responsible for an almost continuous stream of startups, bringing new inventions and discoveries to market at a dizzying pace.
Langer and a former postdoctoral student, David Edwards, now a professor at Harvard, created Advanced Inhalation Research in 1997 to develop pulmonary drug delivery. In 1998, Langer and Jay Vacanti founded Reprogenesis to build polymer scaffolding for regenerative tissue. This technique, once thought controversial, is now the basis of regenerative medicine, which produces artificial skin for patients with severe burns or skin ulcers. The same year, Langer started Sontra Medical (later renamed Echo Therapeutic) to provide transdermal drug delivery.

In 1999, Langer formed MicroCHIPS (with his former graduate student, John T. Santini, Jr.) to develop silicon-chip-based drug delivery, and Transform Pharmaceuticals (later acquired by Johnson & Johnson) for polymorph crystallization. These were followed in the next three years by Combinent Biomedical Systems for transvaginal drug delivery, Pulmatrix for inhaled medication, and Momenta Pharmaceuticals for complex sugar-based therapeutics. Momenta’s sugar-sequencing tools have been used to create more effective blood thinners. In the midst of this success, the FDA finally approved the use of an anti-cancer drug based on the research Langer had first published 28 years earlier, as a postdoctoral student in Judah Folkman’s surgical laboratory.

Meanwhile, Langer and his students continued to break new ground in medical research. In 2004, his new company, Pervasis, introduced new therapeutics for vascular healing. The following year saw the creation of three more businesses based on Langer’s work. Arsenal Medical, which was founded to develop nanofiber-based drug delivery, eventually split into two companies, Arsenal Vascular and 480 Biomedical, which produces resorbable scaffolds for tissue growth. In Vivo Therapeutics produces scaffolds specifically for spinal cord therapy. Not all of Langer’s ventures are concerned with life-threatening disease or crippling injury. Living Proof develops hair and skin care products, including a hair-thickening agent based on a material Langer’s lab originally created to treat prostate and ovarian cancer.

Langer oversaw the creation of three new enterprises in 2006: Semprus BioSciences, for medical device coatings; BIND Biosciences for targeted nanoparticle-based therapeutics; and T2 Biosystems for nanoparticle-based diagnostics. The following year saw the birth of Selecta Biosciences for targeted nanoparticles. In 2008, Langer opened Taris Biomedical for urological drug delivery, and Seventh System Biosystems, which produces a microneedle patch for drawing blood outside of medical facilities. This work, along with a controlled-release polio vaccine developed in the Langer Lab, has been financed by the Bill & Melinda Gates Foundation for use in the developing world. In 2009, Langer and a Johns Hopkins professor (and former Langer student), Justin Hanes, started Kala Pharmaceuticals to provide mucosal drug delivery.

Langer’s latest ventures include Moderna for modified messenger RNA delivery, and Blend Therapeutics, for combination medicines. In 2012, MicroCHIPS completed the first human trials of a wirelessly controlled chip that delivers a drug to stimulate bone formation in osteoporosis patients. A silicon chip with doses of the drug stored in individual reservoirs, each capped with a thin film of platinum and titanium, it releases its contents in response to wireless electric signals. Langer predicts that sensors and drug delivery function will someday be incorporated in a single implantable device which can release drugs in direct response to signals from the body.

That same year, Robert Langer received the Priestley Medal, the highest honor of the American Chemical Society. It was the first time in 65 years that the award was given to a chemical engineer, and the first time it was given to a bioengineer. In 2013, he received the newly created Breakthrough Prize in Life Sciences. Financed by a group of entrepreneurs, including Google’s Sergey Brin and Facebook’s Mark Zuckerberg, the award amounts to $3 million per individual recipient, making it the largest prize purse in science, more than twice as much as that shared by multiple Nobel Prize recipients in any category. In 2015, his accomplishment was honored with the £1 million Queen Elizabeth Prize for Engineering, for his revolutionary leadership “at the interface of chemistry and medicine.”

Today, Robert Langer occupies an unparalleled position in science, business and education. He is the David H. Koch Institute Professor in the Department of Biochemistry. An Institute Professorship is the highest honor a faculty member can hold at MIT; Robert Langer’s professorship was endowed by MIT graduate David H. Koch of Koch Industries. Langer Laboratory at MIT — with over 100 students, postdocs and visiting scientists at any one time — is the world’s largest academic biomedical engineering laboratory.

The Langer Laboratory has served as an incubator for 40 independent companies. Dr. Langer spends roughly one day a week working with the businesses he helped start. His entrepreneurial activities have made him a millionaire many times over, but his primary motivation remains improving the health of men, women, and children around the world. In recent years, Langer Lab has been working with the U.S. Army on a regenerative tissue project for wounded soldiers.

In 2019, researchers from the Langer Lab reported successful animal tests of a contraceptive pill that would only need to be taken once a month, as opposed to the daily dose now required. The Bill and Melinda Gates Foundation is investing $13 million in further development of the monthly pill, which they hope will increase women’s reproductive health options, particularly in developing countries. Dr. Langer has suggested that the same delayed-release mechanism may eventually be applied to treatments for AIDS, Alzheimer’s, opioid addiction and certain mental illnesses.

As of 2020, Robert Langer had authored more than 1,400 publications. More than 1,300 patents had been issued in his name or were pending; more than 400 companies had licensed or sub-licensed Langer Lab patents. His work has been cited in scientific publication more than 304,000 times, more than that of any other engineer in history.

Dr. Langer has licensed or sub-licensed patents to more than 400 biotech, pharmaceutical, chemical and medical companies. In January 2020, one of the companies Langer co-founded, Moderna, began working with the National Institute of Allergy and Infectious Diseases to create a vaccine for the coronavirus Covid-19. In May of that year, Moderna announced preliminary results from the first human study of an experimental vaccine.
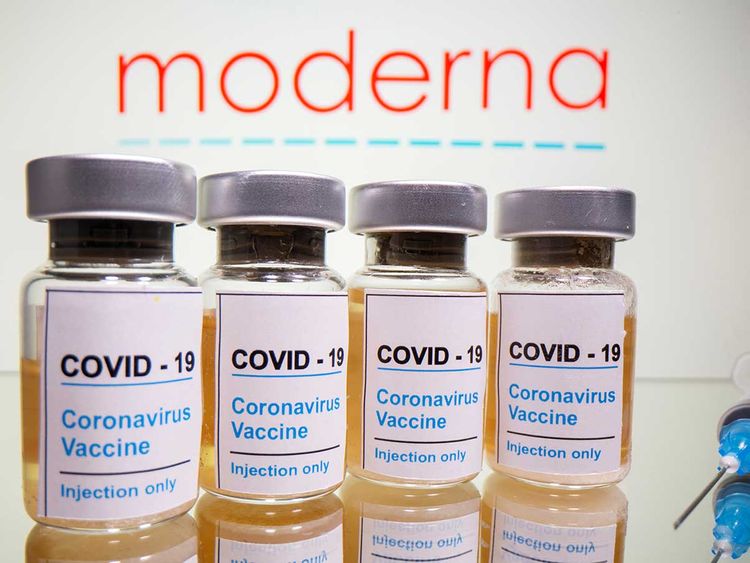
Moderna’s capabilities have come together to allow the authorized use of one of the earliest and most-effective vaccines against the Covid-19 pandemic. The Moderna Covid-19 vaccine became available under an Emergency Use Authorization granted by the FDA on December 18, 2020. Since then, Moderna has released more than 300 million doses of the vaccine to the U.S. government. The Moderna Covid-19 vaccine is one of many drugs in the company’s pipeline.

It is also exploring cancer therapies, cardiovascular blockages, and creating proteins inside organs where they are missing or defective. Moderna is currently developing 25 drug candidates. Moderna has a staggering market value of almost $160 billion and anticipates 2021 revenues of over $20 billion from sales of the vaccine. Moderna’s share price has soared 1,919 percent since 2019.

It has been estimated that the lives of as many as two billion people have been touched by the technologies created by Robert Langer and his research teams. Many of his former students now lead companies or laboratories of their own and advise governments on medical technology. As a colleague has said of Dr. Langer’s students, “They come away thinking that nothing is impossible.”

In January 2022, the BBVA Foundation Frontiers of Knowledge Award in Biology and Biomedicine was given to Robert Langer, Katalin Karikó, and Drew Weissman “for their contributions to messenger RNA (mRNA) therapeutics and delivery technology that enable our own cells to produce proteins for disease protection and treatment.” Langer’s paper, published in Nature in the 1970s, was the first to show it was possible to take nucleic acid molecules – such as RNA – and encapsulate them in nanoparticles for release into the body. This insight enabled “packaging of macromolecule therapeutics including mRNA and delivering them into cells, allowing the cellular translation machinery to synthesize the protein/antigen.”

Robert S. Langer is heralded as one of the most prolific inventors in the history of medicine, as well as the father of controlled drug release and tissue engineering. As a young researcher confronted with the problem of accurate drug delivery, he reversed the usual process of working with existing materials, and custom-designed new polymers (plastics) to release medication at a desired rate. His discoveries have saved countless lives and helped launch a $100-billion-a-year industry that includes products ranging from the nicotine patch to coated cardiovascular stents. His inventions include dime-size implantable wafers for the treatment of cancer, drug release systems operated by remote control, and an implantable “pharmacy on a chip” that releases precise quantities of drugs on a programmable schedule.
Langer pioneered the new field of tissue engineering by growing transplantable tissue in vitro on biodegradable plastic scaffolding. Skin grown using Langer’s principles has been approved by the FDA, cartilage is in clinical trials, and his engineered spinal tissue has been successfully transplanted in laboratory animals. Every year, thousands of Americans die awaiting transplants of organs like the liver, heart and lung, but Robert Langer is creating a future where spare organs can be grown in the laboratory.
Dr. Langer has received the highest professional honors for his work, including the Charles Stark Draper Prize, the National Medal of Science, the National Medal of Technology and Innovation, the Priestley Medal, the Wolf Prize, the £1 million Queen Elizabeth Prize, and the $3 million Breakthrough Prize in Life Sciences. In addition to his endowed chair at MIT, he is a faculty member of the Harvard-MIT Division of Health Sciences and Technology and the David H. Koch Institute for Integrative Cancer Research. His research laboratory at MIT is the largest academic biomedical engineering lab in the world, maintaining about $10 million in annual grants and over 100 researchers.
(Dr. Robert Langer addressed the delegates of the Academy of Achievement at the International Achievement Summit in Washington, D.C. His remarks on that occasion are interspersed with the text of this video interview, conducted that same day.)
Dr. Langer, we’ve asked you to talk about how you became involved in this application of chemical engineering to medicine and biotechnology.
Robert Langer: I graduated in 1974, and I graduated in chemical engineering. So at that time there was this gas shortage, not unlike a few years ago, and what happened was that, just like a few years ago, the prices of gas kept going up, but it was even worse then. I live in Boston, and if you had a car, you actually had to wait in line at the gas station for about two hours to get your car filled up. But the consequence of that is that if you were a chemical engineer you got a lot of job offers. So pretty much every one of my classmates, they went to the oil industry, and so I thought that’s what I should do too. So I applied. You didn’t have to work that hard. I actually got 20 job offers. Four were actually from Exxon alone! And one of them made quite an impression on me. I remember going to Exxon in Baton Rouge, and they had some people who were a few years older than me talk to me about what they were doing, and they were saying, “Boy, if you could just increase the yield of this one petrochemical by .01 percent,” they said, “that would be fantastic.” They said, “That’s worth billions of dollars.” And I remember flying back to Boston that night thinking to myself that I really don’t want to do that. I just thought it was boring. So what did I want to do?
I had spent a lot of my time as a graduate student helping start a school for poor children in Cambridge. Cambridge people may not realize, even though it’s got Harvard and MIT, in the 1970s it had the highest dropout rate in the United States for a city its size. So I got very interested in trying to help this school develop new chemistry and math curricula. I was doing that, and one day I saw an ad in a journal for being assistant professor to develop chemistry curriculum at City College in New York. And I thought, “That’s great! That’s what I want to do.” So I wrote them a letter applying for the job, but they didn’t write me back. But I really liked the idea, so I found all of the ads I could for jobs like that. I found about 40 of them, and I wrote to all of these universities. And actually, none of them wrote me back! So that wasn’t going real well. So I started to think, “What other ways can I use my background in chemical engineering to help people?” I thought about medicine, so I wrote to a lot of hospitals and medical schools, and they didn’t write back either. Then, one of the people in the lab where I was, he said, “Bob, you know, there’s a surgeon in Boston named Judah Folkman.” By the way he was also in the Academy when he was alive. And he said, “There’s a surgeon in Boston named Judah Folkman.” And he said, “Sometimes he hires unusual people.” He thought very highly of Dr. Folkman. I won’t say what he thought about me. So anyhow, when I went, I was actually the only engineer in Boston’s Children’s Hospital.

Just as an aside, I should say it took 28 years — this is sometimes how it works in medicine — from our earliest paper in Science in this area, until the FDA approved the first blood vessel inhibitor. So today, starting in 2004, there are many of these, and these are some of the most widely used drugs in the world for treating cancer, also for treating certain diseases of blindness, like macular degeneration.
I began working on two projects that were actually related. One, to see if we could actually discover the first substance that could stop cancer blood vessels from growing, and hopefully then stop cancer growth. And two, to develop plastic systems — polymer systems — that could slowly release these and other large molecules for a long time, so that we could test these substances. Now, before I started working on this problem, no one had been able to develop ways to continuously release these substances for a long time from bio-compatible polymers, and in fact, if you look in the scientific literature, they said actually it’s impossible to do that, impossible to release these molecules. In fact, really the only thing I had going for me is I hadn’t read that literature! So I actually spent about two years working on the project, and I actually found over 200 different ways to get it to not work. But finally, I made a discovery that I could modify certain types of plastics, and get them to release these molecules over a long time. Then we used these substances to create bioassays that enabled us to discover the first substances that stopped cancer blood vessels, and to help stop cancer.
So about two years after I started working on this polymer delivery project, I got asked to give a talk to a very distinguished audience — to polymer scientists and engineers in Michigan, actually. I had never given a big talk before. In fact, the only talk that I had really given before that one was in eighth grade! That was a minute-and-a-half speech and it didn’t go real well. I remember, the night before that eighth grade speech, I stood in front of my parents’ mirror for about four hours, and I kept reciting it over and over again. And the next day I got asked, when I got up in front of the class, I started reciting it, and I did okay for the first minute and two seconds, but then I couldn’t remember the next word. I just froze. So I stood up there for another minute, until a teacher told me to sit down and gave me a pretty bad grade. I think it was an F. So now, when this big talk in Michigan came many years later, I was very nervous. I kept practicing the talk over and over into a tape recorder. Finally, the day came and I gave it, and I was pretty pleased by the end of the talk this time. I didn’t forget too much what I was going to say, I didn’t stammer too much. So I thought, when I was done with this scientific talk, that all of these older distinguished chemists and engineers being nice people — actually, I should tell you — there’s probably very few scientists in the audience. Because usually when I say that, people know that scientists aren’t always so nice! So I thought they’d actually want to encourage me, this young guy, but actually, when I got done with the talk, a number of these scientists came up and they said, “We don’t believe anything you just said.” They said, “You can’t do that. You can’t get these things through polymers.” And it wasn’t until several years later that people began repeating what we did. The question shifted to, “How could this happen?” I spent a lot of my early career at MIT trying to figure that out.
Shortly after this talk I tried to get grant money to help do different projects. And I remember writing one grant to the NIH on cancer and I got the reviews back, which were really, really negative. They not only didn’t give me the money, they said, “Well, how could Dr. Langer do this?” They said, “He’s a chemical engineer. He doesn’t know anything about biology, and he knows even less about cancer.” I wasn’t sure how that was possible to know less than nothing, but somehow that was the review. Anyhow, also, when I was done with my postdoctoral work I applied for different faculty positions to different chemical engineering departments. But when I did that, I had a lot of trouble getting a job, because they all felt I wasn’t doing engineering. They felt it was more biology. So I ended up joining what was called the Nutrition and Food Science Department at MIT. But in that department what happened was, about a year after I got the position, the chairman of the department left. So then a number of the senior members of the department decided they’d give me advice, and their advice was I should start looking for a new job. So there I was. I was getting my grants turned down, people didn’t believe in my research, it appeared like I wasn’t even going to get to be an associate professor. But fortunately, what happened over the next few years is different people in academics and in pharmaceutical companies started using a lot of our principles, and things slowly began to turn around.
I wanted to have our inventions to get to the point where they could really help people, and it’s very difficult, because it takes a lot of money to do that. So I began writing patents on our work, and today we’ve licensed or sublicensed these patents to actually 250 different companies. Actually, with my students, we even started about 25 companies. I should add that when I first started doing this as a professor in the early ’80s, most academic scientists looked down upon it. They said professors shouldn’t do things like that. But today these companies have made all kinds of products that treat patients — and I’ll mention some of this — with cancer, heart disease, and many other sicknesses, they’ve created tens of thousands of jobs.
One of the big challenges is getting a patent. That’s actually key in a lot of scientific innovation. So we actually filed the first patent in the history of Boston’s Children’s Hospital in 1976, and the patent office turned it down. They actually turned it down five times between 1976 and 1981, and my lawyer just said we should give up, but I don’t like to give up that easily. So I started thinking about other ways, not even scientific ways, that we could get the patent allowed, nothing illegal. But anyhow, so what the patent examiner said — because he didn’t understand the science that well — is what we did was obvious. So I thought, “How could he think that?” because every time I gave a talk on this everybody told me it was impossible, it couldn’t work. I wondered whether scientists had ever written that down — not just insult me to my face — whether they’d actually ever even written it down. So I scoured the literature to see what anybody had written on me. And actually I found an article in 1979, a number of years after we wrote our article, that actually was very interesting, because they referred to our work and they wrote — and I’ll just give you a quote — they said, “Folkman and Langer…” myself, “…have reported some surprising results…” They used those words, “…that clearly demonstrate the opposite of what scientists had thought before.” So I showed that quote to our lawyer, and he said, “That’s very interesting.” He said, “I’m going to fly down and talk to the patent examiner.” And the patent examiner said to the lawyer, he said, “You know,” he said, “I didn’t realize that.” He said, “If Dr. Langer can get affidavits from the five people who wrote that quote, that they really wrote that quote, I will allow that patent.” So I wrote them, and all five of them were nice enough to say that they really wrote it! And we got the patent.
I was pretty naive about this. I had worked on this for nine or ten years. Nobody was using it, but then one company and then a second company — one in animal health and one in human health — wanted to license it, and they actually gave me a consulting fee, and grant money, and I was so excited about this I got a better car, and I had money to support our lab. But these companies were very large. They were multibillion-dollar companies. And what happened was — even though they gave us grant money at MIT — that what happened is, they themselves would do maybe one or two experiments over a year or two, and if they didn’t work out right — and in science most things don’t work out right the first time — they just gave up. So a few years later, one of my friends at MIT, Alex Klibanov, he said, “Bob, why don’t we just start a company ourselves?” So I was able to get these patents back, and we started a little company, Enzytech, and that later merged with our downstairs neighbor to become Alkermes. They used these microspheres to develop drug delivery systems for all kinds of drugs.

Some of them can treat cancer. One of them has actually been used in over 5 million schizophrenic patients. There are ones for treating alcoholism and narcotic addiction. Many of these are now on the market that have affected many millions of people. To move to the future of some of the things we’re doing now, we can use a lot of these same principles, not only to create little microspheres, but even nanoparticles that can zoom in and hopefully someday provide a new way of thinking about treating cancer and other diseases.


